|
INTRODUCTION | ORIGINAL STUDIES | THIS STUDY | RESULTS | BOTTOM LINE | COMMENTS |
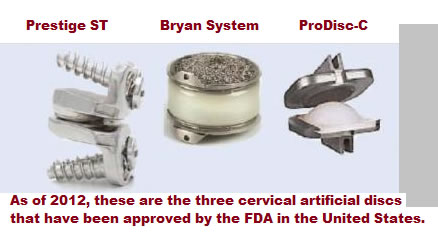
Upadhyaya CD, et al. "Analysis of the Three United States Food and Drug Administration investigational device exemption cervical arthroplasty trials." Spine-2011; DOI:10,3171/
 |
I have been waiting for the publication of this investigation for a long time. At last we finally have a very well executed, independent investigation (the two lead-researchers were not associated with the manufacturers of the artificial discs investigated and declared no conflicts of interest) into the efficacy and safety of cervical artificial disc implantation (arthroplasty) for the treatment of chronic neck pain, radiating upper limb pain, and/or extremity dysfunction (myelopathy).
TERMINOLOGY: some of the terminology can get confusing when it comes to artificial discs and techniques for implanting them. Arthrodesis basically means traditional fusion--you are basically making a joint immobile. |
An example would be anterior cervical discectomy fusion (ACDF). Arthroplasty is a term that means something is being implanted into the joint that allows motion. A perfect example is artificial disc implantation – this is an arthroplasty.
Throughout this webpage, I will use ACDF and arthrodesis synonymously. I will also use artificial disc implantation and arthroplasty synonymously.
INTRODUCTION:
Cervical disc-herniation-induced neck pain and radiculopathy stricken thousands of Americans each year and costs millions of dollars in medical and hospital fees for service. The "gold standard" treatment for such a condition had been the time-tested anterior cervical discectomy and fusion (ACDF), which boasts a 95% fusion/stabilization rate and high patient satisfaction [2,13,22]. The Major drawback to ACDF stems from a biomechanical change in the cervical spine following the fusion/immobilization of the target vertebrae. More specifically, the workload (biomechanical stress) upon the above and below the fused segments becomes dramatically increased, which in turn may lead to failure of the disc above and/or below the site of original surgery. This phenomenon, called the "dominoes effect," often necessitates a second surgical procedure in order to correct the new problem.
Cervical arthroplasty was developed as an alternative to ACDF. Although cervical arthroplasty and ACDF both remove and stabilize the damaged disc and their respective vertebral segments, cervical arthroplasty (implanting the cervical artificial disc) does so without hindering the normal motion at the level of implantation. Therefore, it was theorized that by preserving the intersegmental motion of the operated cervical spine, there would be a resultant elimination of the dominoes effect , which in turn would lead to a decrease in the need for second adjacent level surgery.
ORIGINAL STUDIES:
In general, each of the three artificial disc trials compared their product against ACDF, for the treatment of patients that suffered severe unrelenting (intractable) neck pain, radiating arm pain (radiculopathy) and/or neurological dysfunction (i.e., trouble with movement of the hands, arms, and/or legs [myelopathy] secondary to single level compressive degenerative change and/or disc herniation.
All three trials were constructed with similar inclusion and exclusion criteria as well as used similar clinical outcome measures (Neck Disability Index, SF-36, and patient pain scores [VAS and 0--10]), which were used to assess the outcome of the surgery. Patients were then followed for a minimum of 24 months and then called in for re-examination and assessment.
Each of these three trials consisted of a large sample size. That is, there were over 100 patients in each study arm. Such large experimental groups (cohorts) give rise to excellent clinical data and were in fact mandated by the FDA for this type of investigation. Another fine and necessary achievement of these three trials was the number of patients available at two-year follow-up examination. More specifically, approximately 90% of the patients (approximately 94% for the arthroplasty group and 87% for the ACDF group) were available for re-examination at the two-year mark-- this is statistically powerful.
Exclusions/Inclusions:
All three studies excluded patients that had multi-level cervical spondylosis, evidence of cervical instability, single level severe stenosis, target segment that severely lacked motion, metabolic bone disease, and/or a target segment that had lost 50% or more of its height. [Comment: undoubtedly, this excluded quite a few of the real world patients.]
Inclusion criteria for the study was also similar between the three artificial disc studies: the Prestige ST study included patients that suffered single level spondylosis in one of the bottom five cervical vertebrae who presented with intractable radiculopathy and/or myelopathy. The Bryan study included patients with cervical disc herniation in one of the bottom five cervical discs with resultant intractable radiculopathy or myelopathy. The ProDisc-C study included patients with symptomatic spondylosis in one of the lower five cervical discs, which resulted in intractable radiculopathy, myelopathy, and neck pain.
THIS STUDY:
In this 2011 investigation, a group of exceptionally bright researchers/statisticians took a closer look at the data (some of which had never been published before) behind the three recently published artificial disc studies (see above). The object of this study was to combine the data from all artificial disc trials in order to achieve higher statistical power and better to better control study variations. In other words, they wanted to independently confirm that these three cervical artificial discs were in fact a safe and afforded an effective alternative to traditional ACDF.
 |
The manufacturers were apparently receptive to this scrutinization, for they not only volunteered all the data from their investigations (some of which was never before published), but they also loaned the investigators of this paper some of their own investigators who had in fact participated in the original artificial disc studies. Noteworthy is the fact, however, that none of the "loaned" coinvestigators were allowed to extract the data for pooling in this investigation.
The image to the left is of an early generation of the Prestige artificial disc system. |
The data scores extracted from the three investigations, which included data from the neck disability Index, SF-36 (both the MCS and the PCS), as well as pain scores in the form of visual analog scores and a simple 0-10 format.
All in all, a total of 1213 patients (621 in the arthroplasty groups and 592 in the ACDF groups) were analyzed in this investigation. The average age of patients was approximately 44 years old.
Confounding Groups:
It is known that patients involved in the litigation process (i.e., workers compensation and personal injury patients) can skew the outcome of investigations because of ulterior motives to report improvement. Wisely, the three artificial disc trials kept these types of patients to a minimum.
More specifically, the total combined sample size of patients involved in litigation was approximately 16% (10% workers compensation patients and 6% personal injury patients). Unfortunately, they did not single out this sample like some researchers decide to do.
Neurological success scores, as well as surgical outcome data were also combined and evaluated. However, the investigators were unable to successfully combine the "adverse events" and "return to work" data because of severe heterogeneity between the three studies.
RESULT SECTION:
NECK DISABILITY INDEX | PAIN SCORES | X-RAY MOTION | REVISION SURGERY | ADJACENT LEVEL SURGERY | NEUROLOGICAL SUCCESS | TO THE TOP |
Neck Disability Index (NDI): [back to results]
I chose to report on the NDI because this is an extremely common methodology that has been used for years in the healthcare industry for assessing patient condition. As you go over the next three graphs, you will notice that between 30%-40% improvement can be anticipated with regard to pain and function with either treatment option.




CONFOUNDERS:
There is one strange finding in the data (the research team failed to comment on this) that certainly raises an eyebrow. It had to do with return to work. More specifically, the Prestige ST and Bryan studies both reported that approximately 15% of the patients who were not working at the start of the study, were in fact able to return to work at the two-year mark. On the other hand, the ProDisc-C study could boast no such results. That is, only 1% of the arthroplasty patients, who were not working at the start of the study, were in fact able to return to work at the two-year mark. The ACDF portion of that study was even worse: at the end of two years, -6% of them were working (i.e. nobody return to work and more were out of work).
Perhaps this had to do with the recession and the geographical location of these patients. But whatever the cause, somebody should have commented on that.

Pain Scores: [back to results]
With regard to neck and arm pain and intensity, all patients in all groups demonstrated good improvement over that two-year interval. Although there was no statistical difference, the ACDF patients reported a greater improvement from their neck pain. More explicitly, on average the ACDF patients improved 37.26%, while the artificial disc group improved 32.38%.



Radiographic Outcome--Was The Motion Preserved? [back to results]
The whole purpose of designing the artificial disc was to preserve the biomechanics of the cervical spine following surgical removal of a symptomatic, degenerative disc. So naturally, before and after radiographs were taken in all three artificial disc studies.
In all three artificial disc studies, the goal was accomplished: there was good preservation of cervical spine range of motion at the target segment and all artificial discs. In fact, when compared to the preop radiographs, the Post-Op radiographs demonstrated even greater motion at the level of the artificial disc.
So the next burning question is whether or not the preserved movement (the goal of artificial disc implantation) resulted in a decreased need for second surgery.
Adjacent Level Surgery: [back to results]
So do these prosthetics cervical discs work? I mean, do they help prevent the domino effect and second surgery? According to the pooled data, it was a very close call at the two-year mark (which honestly is a little premature to assess this finding). That is, there was a statistical difference favoring the artificial discs effectiveness for preventing adjacent level surgery when the numbers were run through the fixed effects model. However, using the more conservative model of statistics (i.e., the random effects model), the artificial discs failed to prevent the domino effect and second adjacent level surgery. The data is coming below, but note the variation-- that's why it was a great idea to pool all the data!



Revision Surgery (The first surgery didn't work!) [back to results]
*There was a significant finding with regard to second surgeries. More specifically, patients who underwent ACDF had a statistically confirmed increase in the number of required second surgeries. Second surgeries for this paper were described as revision surgery, supplemental fixation, and reoperation (not including adjacent segment surgery). This was confirmed by both fixed effects analysis, as well as random effects analysis.
Neurological Success--Little Misleading:
First of all, what is neurological success? It was defined as an improvement or *non-worsening of the patient's neurological condition following surgery. Anyway, a patient was deemed to have a "neurological condition" if he/she had any abnormality upon motor, sensory, and/or reflex examination.
The results of this assessment revealed that only arthroplasty (artificial disc implantation) was statistically successful at improving or maintaining the patient's presurgery condition. That is, the artificial discs did better at fixing/maintaining any patient neurological symptoms when compared to ACDF.
Comment: I thought it was rather strange and even misleading that all of the papers (even this one) allowed "neurological success" to be defined as a non-worsening of an already present neurological condition, which of course was caused by the problem (well, at least one of the problems) these devices were trying to fix. Part of the goal of either surgery is to try to rid the patient of neurological symptomatology, so in my opinion no improvement of a patient's neurological status should have been a failure–But it wasn't. Unfortunately, these investigators did not put forth the data of patients who actually improved following surgery with regard to the neurological status-- that would've been more useful.
THE BOTTOM LINE:
 |
This was an excellent investigation and the researchers are to be commended for publishing it. When trying to predict the safety and efficacy of a new procedure’s effect upon the general public, the more experimental sampling you have from the target population, the more confidence you will have in the outcome of that study. In other words, you can be more certain that the treatment works. |
In this case, the researchers combined the data from three multi-center, noninferiority, prospective, randomized-controlled US FDA-approved ID investigations in order assess the combined efficacy of the three artificial discs devices (arthrodeses) recently approved by the FDA for the treatment of single level disc disease in the cervical spine.
The bottom line is that this statistically powerful investigation (more powerful than the original studies that led to FDA approval of each respective artificial disc) confirmed that the cervical artificial discs are just as effective as the gold standard treatment (anterior cervical discectomy fusion [ACDF]) for the treatment of single-level symptomatic cervical disc herniation (with or without spondylosis/degenerative change), which have resulted in intractable neck pain, arm pain, and/or neurological loss of extremity function (myelopathy).
Arthroplasty Even Better then arthrodesis: although the study design (noninferiority) was geared to demonstrate that arthroplasty was just as effective as ACDF, there were three outcomes that were statistically powerful enough to suggest that arthroplasty (the cervical artificial disc surgery) was even more successful than traditional ACDF:
(1) Revision Surgery: There was strong statistical evidence to suggest that the cervical artificial discs lessened the surgical complication rate and need for second, revision surgery. That is, patients who had the cervical artificial discs implanted had a lesser chance of that procedure failing, which in turn would result in the need for revision surgery, when compared to patients that underwent ACDF.
(2) Neurological Success: There was strong statistical evidence that the cervical artificial discs had a better chance of ameliorating or stabilizing the patient's neurological dysfunction. More specifically, patients who had the arthroplasty, had either an improvement or a stabilization of neurological symptomatology such as diminished/lost deep tendon reflexes, sensory abnormalities (numbness, tingling, burning), and/or motor abnormalities (muscle weakness and/or atrophy).
(3) Adjacent Level Disease: There was mild statistical evidence (only one of the two statistical methodologies demonstrated success) that the cervical artificial discs reduced occurrence of the nemesis of ACDF surgery: adjacent level disease. Really the whole purpose for inventing the cervical artificial disc was to prevent something called the "dominant effect." The dominoes effect occurs after ACDF because two vertebrae are fused together, which in turn disrupts the biomechanics of the cervical spine. That is, this situation places more load/stress on the disc and vertebra above and below the level of ACDF, which in turn can result in adjacent level degenerative disc disease, which in turn can lead to disc herniation induced neck pain, radiculopathy, and/or myelopathy, which in turn can lead to the necessity for a second surgery.
With regard to preserving intersegmental motion (at least with regard to flexion and extension), all cervical three artificial discs did there thing. That is, at the two-year mark all operated segments had excellent movement as demonstrated on flexion/extension radiographs. Of course the ACDF patients demonstrated no movement.
Comment: in my opinion it is really too early to tell whether or not arthroplasty prevents the domino effect. Empirically speaking, adjacent level disease secondary to ACDF rears its ugly head at approximately the 5-year time point. So hopefully this group of researchers (or somebody) will reassess this cohort at five years.
COMMENTS:
 |
Hats off to this research team, who double-checked and confirmed (in a more statistically powerful manner) the previously-published manufacturer investigations that led to the recent FDA approval for the first three cervical artificial discs (Prestige ST, Bryan, and ProDisc-C), which may now be used to treat the general public for symptomatic single-level cervical disc disease. |
Without question, the cervical artificial discs have preserved the biomechanics of the cervical spine at the two-year follow-up, although the jury is still out with regard to whether or not that preservation pays off by reducing the need for adjacent level surgery in the future. I eagerly await the five-year investigation, which should tell the complete story.
But regardless of whether or not the cervical artificial discs do in fact prevent future adjacent level surgery, there are already statistically powerful advantages to choosing arthroplasty over ACDF. More specifically, with arthroplasty the patient has a better chance at preventing revision surgery (the first surgery failed and needs fixing) and has a better chance of either improving or stabilizing there neurological symptomatology.
Another thing to keep in mind is surgeon skill: lumbar fusions are typically performed posteriorly (from the backside), which puts them behind the learning curve (compared to surgeons from Germany, for example) with regard to implanting artificial discs anteriorly into the lumbar spine.
With regard to the cervical spine, however, US surgeons are very skilled at this anterior approach (ACDF is done from the anterior) and should have no surgical learning curve with regard approaching the cervical spine from the anterior – a big plus. So the learning curve is greatly reduced to only the new technique of implantation.
Therefore, I am much less hesitant to (in my layperson opinion) recommend considering cervical artificial disc surgery over anterior cervical discectomy fusion for patients of home match the study entry criteria (single level symptomatic cervical disc herniation with or without spondylosis).
ANOTHER STUDY: There was another investigation (a META study) that basically concluded the same thing: the cervical artificial discs are as effective as ACDF.[here] McAfee et al. Spine (2011) Oct 27. This one also added another FDA IDE study that was not covered above--i.e., the Porous Coated Motion (PCM) cervical disc replacement. (read about the trail here).
[other comments: | adjacent level surgery | misleading definition | confounding groups |
| Top of the Page |
© Copyright 2002 – 20012 by Dr. Douglas M. Gillard, DC - All rights reserved
|

